Selecting the appropriate reporting guideline for your article
Authors of research articles frequently forget to report details about their study which are important for readers to know. This can delay publication and stop their work being used, cited or replicated. This is a waste of the human and financial resources invested in the research.
Reporting Guidelines and checklists have been developed for a wide variety of research types and study designs which set up the most important things readers need to know about your work.
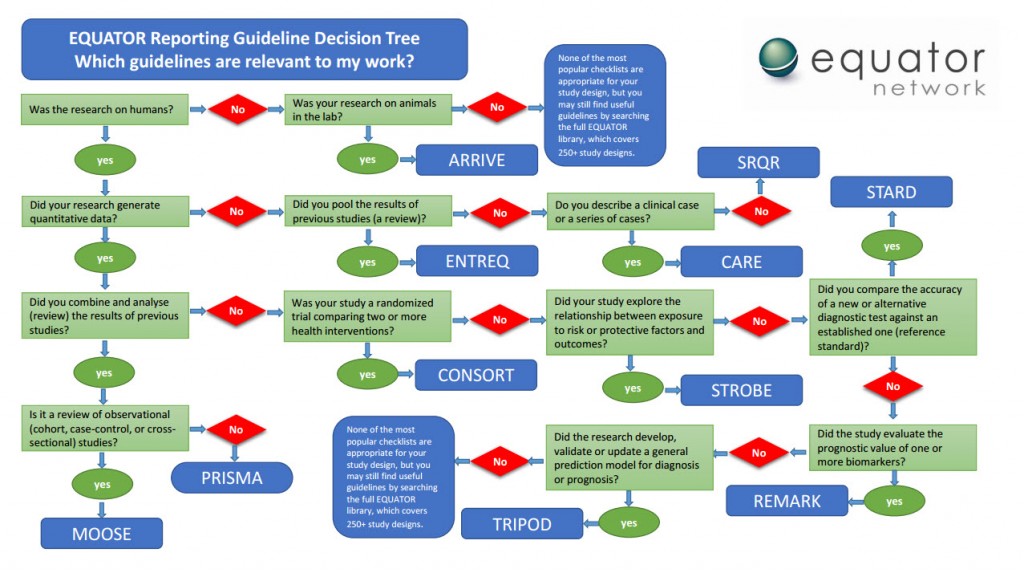
In 2015, EQUATOR created a simple flow chart (PDF) to help authors, editors and peer reviewers find the most appropriate checklist and reporting guideline.
We collaborated with Penelope Research to develop an online wizard based on this chart. The first version of the EQUATOR wizard was piloted by BioMed Central in February 2016. It was embedded into the submission system for four BMC journals and the promising results of a before-and-after analysis were published in Research Integrity and Peer Review in December 2017.
Early in 2018, we launched a new version of the wizard called “GoodReports” (www.goodreports.org) which now includes 16 of the most commonly used reporting guidelines. It also directs you to an online fillable version of the checklist to submit to a journal alongside your manuscript. In October 2019 we started a three-year project to improve GoodReports further by adding more reporting guidelines and facilitating the use of more than one reporting guideline where appropriate.
This free tool will help you work out which reporting guidelines are right for you
You can also search the EQUATOR Database of Reporting Guidelines, or read more about different study types on the website of Oxford’s Centre for Evidence-based Medicine.